The absence of a coherent federal framework for finished cannabinoid products stems from FDA's failure to implement rules under DSHEA and DEA's narrow interpretation of the Controlled Substances Act. While the 2018 Farm Bill legalized hemp cultivation, no coherent framework exists for post-harvest manufacturing and retail cannabinoid products.
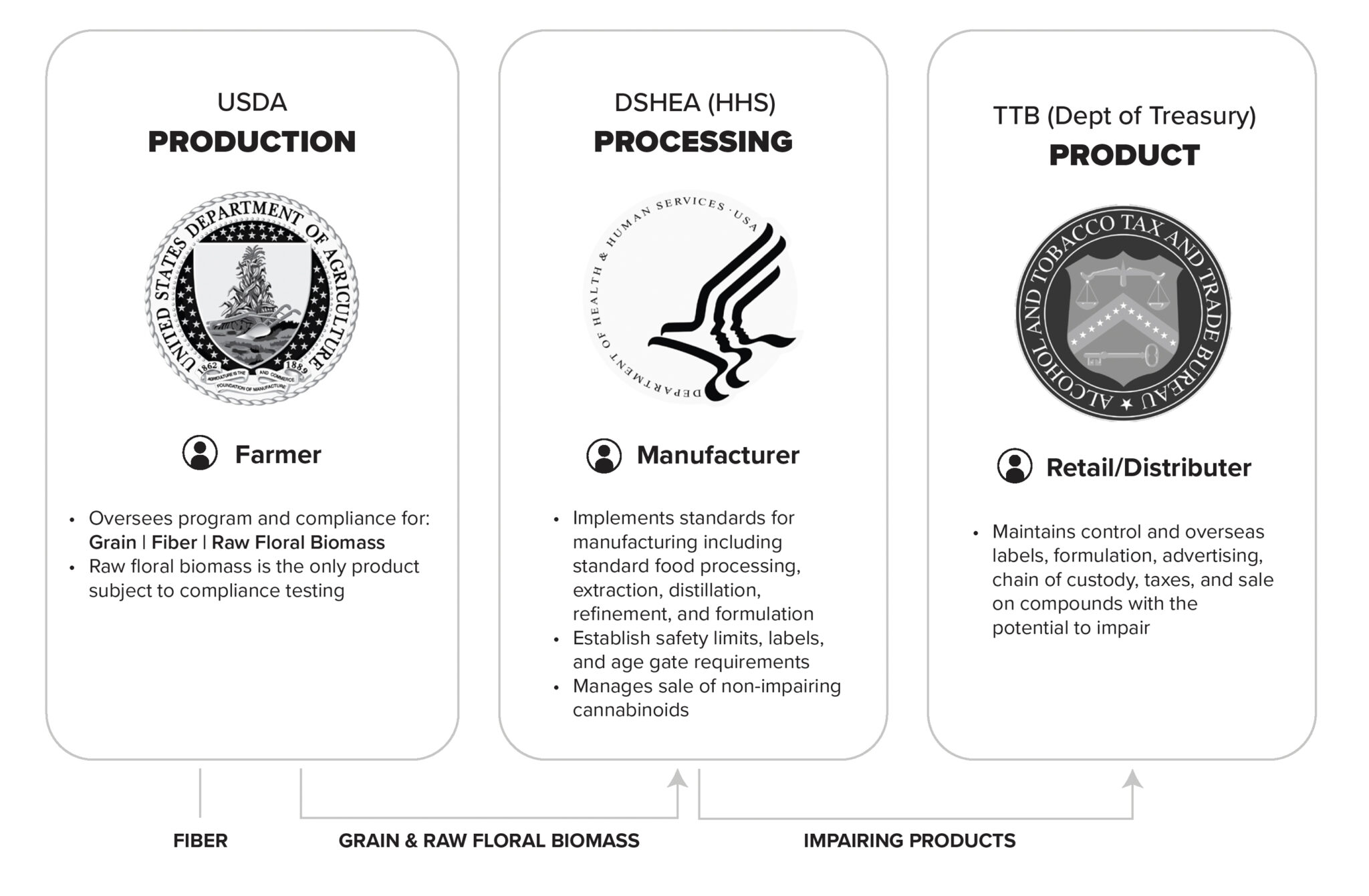
The HEMI Working Group proposes bifurcating industrial hemp (fiber/grain) from floral hemp (cannabinoid-producing), with regulatory oversight distributed across USDA, FDA, and TTB.
Three-Step Policy Framework
Step 1 — Farm Bill
Establish separate definitions for industrial and floral hemp. Increase total THC threshold from 0.3% to 1.0% and clarify USDA's agricultural jurisdiction.
Step 2 — Energy & Commerce
Direct FDA to regulate non-impairing cannabinoids through DSHEA amendments and maintain prior GRAS determinations.
Step 3 — Energy & Commerce
Assign TTB responsibility for potentially impairing cannabinoid products modeled on alcohol regulation, including age restrictions and excise taxes.
Intended Outcome
The framework aims to align agricultural oversight (USDA) with public health regulation (FDA, TTB, DEA) and eliminate regulatory gaps post-Loper Bright.
Download the Full Policy Document (PDF)